The effects of pretreatment with nalxonaxine (NLXZ), a μ1 opi- ate receptor antagonist, and naltrindole (NTI), a δ opiate receptor antagonist, on 7-HMG and morphine self- administration were also examined. Both NLXZ and NTI reduced 7-HMG self-administration, whereas only NLXZ de- creased morphine intake. The present results are the first to demonstrate that 7-HMG is readily self-administered, and the reinforcing effects of 7-HMG are mediated in part by μ and δ opiate receptors. In addition, prior exposure to 7-HMG increased subsequent morphine intake whereas prior exposure to MG decreased morphine intake. The present findings indicate that MG does not have abuse potential and reduces morphine intake, desired characteristics of candidate phar- macotherapies for opiate addiction and withdrawal, whereas 7-HMG should be considered a kratom constituent with high abuse potential that may also increase the intake of other opiates.
Introduction
The increasing use of kratom (Mitragyna speciosa korth; aka thang, kakuam, thom, ketum, biak biak), a plant indig- enous to Southeast Asia, has emerged as a public health concern in the US. Kratom has been used traditionally to combat fatigue and increase work productivity amongst farm populations in Southeast Asia. Kratom leaves are chewed or made into an extract and brewed (Hassan et al. 2013; Warner, Kaufman, & Grundmann 2016), and consumption is reported to produce stimulation (at low doses) and opiate-like effects (at higher doses) including analgesia, antitussive, antidiarrheal, and anti-inflammatory
effects.
Historically, kratom has also been used to reduce the intensity and duration of opiate withdrawal symp- toms (Boyer et al. 2008; Ward et al. 2011; Cinosi et al. 2015; Warner et al. 2016); however, studies assessing the clinical efficacy of kratom are limited. In Southeast Asia, regular kratom use has been associated with phys- ical dependence and withdrawal symptoms (Suwanlert 1975; Saingam et al. 2013; Singh, Muller, & Vicknasingam 2014), effects attributed in large part to mitragynine (MG). In the United States, there is growing concern regarding the safe use of kratom based on reports of addiction (Sheleg & Collins 2011; Galbis-Reig 2016) and toxicity and fatalities associated with use (McWhirter & Morris 2010; Neerman, Frost, & Deking 2013; Singh, Narayanan, & Vicknasingam 2016; Drago et al. 2017; Fluyau & Revadigar 2017).
Various strains of kratom are widely available over the internet as well as in various locations throughout the country. Forecasting models indicate kratom use will con- tinue to increase in the United States (Stogner 2015). Currently, kratom consumption remains legal in the ma- jority of states in the US. FDA’s associate commissioner for regulatory affairs has stated that the FDA has ‘identified Kratom as a botanical substance that poses a risk to pub- lic health and has the potential for abuse’ (Food and Drug Administration, 2016). Concerns about Kratom have led the FDA to issue an import alert and public health advi- sory and the DEA to include kratom on the list of Drugs and Chemicals of Concern.
Related: Kratom Legality Map and information. States, Cities and Counties where Kratom products are prohibited
Kratom leaves contain more than 25 identified alkaloids (Hassan et al. 2013). Mitragynine (MG) and 7- hydroxymitrgynine (7-HMG), the main psychoactive alkaloids of kratom, constitute approximately 60 and 2 percent of the plant’s alkaloids, respectively (Prozialeck, Jivan, & Andurkar 2012). MG and 7-HMG are partial agonists at the μ opiate receptor and weak antagonists at δ and κ opiate receptors (Kruegel et al. 2016; Varadi et al. 2016), with 7-HMG exhibiting approximately 5-fold greater affinity at the μ opiate receptor compared to MG. Assays of opiate receptor-mediated G-protein function re- veal similar potencies for MG and 7-HMG (Kruegel et al. 2016; Varadi et al. 2016). While both compounds exhibit naloxone-sensitive antinociceptive activity, 7-HMG exhibits 40-fold greater potency than MG and 10-fold greater potency than morphine in these assays (Takayama et al. 2002; Matsumoto et al. 2004). Repeated administration of 7-HMG produces tolerance to the compound’s analgesic effects as well as cross-tolerance to morphine’s antinociceptive action (Matsumoto et al. 2005). Chronic consumption of kratom as well as repeated administration of 7-HMG induces physical dependence as determined by naloxone-precipitated withdrawal (Matsumoto et al. 2005).
Few studies have examined the abuse/addiction po- tential of MG and 7-HMG. The absence of controlled stud- ies in humans creates space for basic science studies to provide critical information to help guide use and policy decisions. The abuse liability of compounds is generally assessed in animal models using drug discrimination, place conditioning and/or self-administration paradigms —all of which address different aspects of abuse. Studies in animal models have provided considerable insight into the behavioral effects of MG and 7-HMG as well as the po- tential neurobiological mechanisms underlying those ef- fects. Acute administration of MG increases locomotor activity, induces anxiolytic effects and induces condi- tioned place preference (Yusoff et al. 2016), while re- peated MG administration induces locomotor
sensitization but impairs performance on a variety of cognitive tasks (Yusoff et al. 2016; Ismail et al. 2017). Moreover, MG and 7-HMG fully generalize to the discrim- inative stimulus effects of morphine, suggesting the potential for abuse (Harun et al., 2015).
The reinforcing effects of drugs are an important indi- cator of abuse liability which is typically evaluated using drug self-administration procedures. Inherent in the op- erational definition of reinforcement is the contingency between behavior and drug administration, an important differentiation between self-administration and place conditioning and drug discrimination. Drug self- administration is widely accepted as the gold standard of measurements for abuse liability (Katz 1989; Hemby 1999; Lynch & Hemby 2011). In spite of the concern of the potential abuse liability of MG and 7-HMG in humans, no published studies to date have examined the ability of these compounds to maintain self- administration in experimental subjects. To that end, we assessed that ability of MG and 7- HMG to substitute for morphine self-administration and to engender and main- tain self-administration in drug naïve animals. Addition- ally, the contribution of μ and δ opiate receptors on the reinforcing effects of 7-HMG was examined. Results of these studies provide an objective assessment of the abuse potential of MG and 7-HMG in a rodent model that reca- pitulates key features of human drug taking.
MATERIALS AND METHODS
Subjects
Male Fischer 344 rats (100–130 days; Charles River, Wil- mington, MA) were housed in a temperature-controlled vivarium on a 12-hour reversed light/dark cycle (lights on at 6:00 PM). Rats were group-housed two per cage with water available ad libitum. Food was restricted such that rats were maintained at 90 percent of their free feed- ing weight throughout the experiment. Experimental ses- sions were conducted during the dark phase of the light/dark cycle. All procedures were performed in accor- dance with the High Point University Institutional Animal Care and Use Committee and the National Institutes of Health Guide for the Care and Use of Laboratory Animals (NIH Publication No. 80-23) revised in 1996.
Chemicals
Mitragynine was isolated according to published proce- dures (Ponglux et al. 1994). 7-HMG was synthesized from mitragynine by McCurdy’s research group, Department of BioMolecular Sciences, University of Mississippi, as previ- ously published (Ponglux et al. 1994; Takayama et al. 2002). Mitragynine and 7-HMG were analyzed by 1H NMR, 13C NMR, elemental analysis, HPLC and HR-MS, and were found to be >99 percent pure. Morphine SO4 was purchased from Gallipot, Inc. (St. Paul, MN), penicillin G procaine from Butler Company (Columbus, OH), propofol, ketamine HCl (Ketaset), xylazine (Xylamed) from Patterson Veterinary Supply, Inc. (Greely, CO), naltrindole and naloxonazine from Tocris Bioscience/Biotechne (Minneapolis, MN). Drugs were dissolved in heparinized saline. Morphine, MG and 7-HMG were infused in a volume of 200 μl.
Behavioral apparatus and training
Operant apparatus
Experiments were conducted in operant conditioning chambers (ENV-008CT; Med Associates, St. Albans, VT) enclosed in sound-attenuating cubicles (ENV-018; Med Associates). The front panel of the operant chambers contained a response lever (4 cm above the floor and 3 cm from the side wall), a cue light (3 cm above the lever) and a food chute centered on the front wall (2 cm above the floor) that was connected to a food pellet dispenser (ENV-023; Med Associates) located behind the front wall and a tone generator to mask extraneous noise. A syringe pump (PHM-100; Med Associates) holding a 20-ml syringe delivered infusions. A counter- balanced arm containing the single channel liquid swivel was located 8–8.5 cm above the chamber and attached to the outside of the front panel. An IBM compatible computer was used for session programming and data collection (Med Associates Inc., East Fairfield, VT).
Lever training
Subjects were transferred to the operant chambers for daily experimental sessions, and responding was engen- dered and maintained by delivery of food pellets (45-mg pellets; Noyes, Lancaster, NH) under an FR 1 schedule of reinforcement that was gradually increased to FR3 (every third response produced a food pellet). The lever light was illuminated when the schedule was in effect. Completion of the response requirement extinguished lights, delivered food and was followed by a 20-second timeout (TO) period during which all lights were extinguished, and responses had no scheduled consequences. After the TO, the lights were illuminated, and the FR schedule was again in effect. Sessions lasted 20 minutes or until 30 food pellets were delivered. Responding was considered stable when there was less than 10 percent variation in the number of rein- forcers for three consecutive sessions.
Intravenous jugular surgery
After operant responding was acquired and maintained by food, subjects surgically implanted with an intravenous jugular catheter. Venous catheters were inserted into the right jugular vein following administra- tion of ketamine (90 mg/kg; IP) and xylazine (5 mg/kg; IP) for anesthesia as described previously (Pattison et al. 2012; Pattison et al. 2014; McIntosh et al. 2015). Cathe- ters were anchored to muscle near the point of entry into the vein. The distal end of the catheter was guided subcu- taneously to exit above the scapulae through a Teflon shoulder harness. The harness provided a point of attach- ment for a spring leash connected to a single-channel fluid swivel at the opposing end. The catheter was threaded through the leash and attached to the swivel. The other end of the swivel was connected to a syringe (for saline and drug delivery) mounted on a syringe pump. Rats were administered penicillin G procaine (75 000 units in 0.25 ml, i.m.) and allowed a minimum of 5 days to recover before self-administration studies were initiated. Hourly infusions of heparinized saline were administered through the catheter to maintain functional catheters. The health of the rats was moni- tored daily by the experimenters and weekly by institu- tional veterinarians per the guidelines issued by the Institutional Animal Care and Use Committee and the National Institutes of Health. Infusions of propofol (6 mg/kg; i.v.) were manually administered as needed to assess catheter patency.
Rats were transferred to the operant chambers for daily two-hour self-administration sessions. Before each session, the swivel and catheter were flushed with 500 μl of heparinized saline before connecting the cathe- ter to the syringe via a 20 ga luer hub and 28 ga male connector. The start of each session was indicated by the illumination of the house light, stimulus light above the lever and the extension of the lever. Completion of the response requirement was followed by a 20-second time out (FR3:TO 20 seconds) during which time the subject received a 200-μl intravenous infusion over the first 6 seconds, retraction of the lever, extinguishing of le- ver light, generation of a tone and illumination of the house light. At the end of the TO, the lever was extended, lever light illuminated, tone silenced and the house light extinguished (Hemby, Smith, & Dworkin 1996; Hemby et al. 1999; McIntosh et al. 2015).
Experiment 1: Substitution for morphine self-administration
Self-administration was engendered using 100 μg/inf of morphine sulfate. When responding was stable (two con- secutive sessions in which the number of reinforcers did not vary by more than 20 percent), the dose was changed to 50 μg/inf. Following stable responding at this dose, saline was substituted for morphine until responding stabilized. Following the conclusion of extinc- tion testing, rats were assigned to one of two groups to
receive MG (n = 9; 25, 50, 100 and 150 μg/inf) or 7-HMG (n = 8; 2.5, 5, 10 and 20 μg/inf)—doses were randomized. The dose range for MG self-administration was based on the finding that equivalent doses of mor- phine and MG induced place conditioning in rats (Yusoff et al. 2016). The dose range for 7-HMG was based on the finding that 7-HMG substitutes for morphine in the drug discrimination procedure at a dose five-fold lower than the training dose of morphine. Once responding stabilized for a particular dose, the next dose was made available the following session until all doses mentioned above were assessed. After all doses had been assessed for a sub- ject within a group, the rat was allowed to self-administer morphine (50 and 100 μg/inf) to determine the effect of prior drug history on subsequent morphine intake.
Experiment 2: Acquisition of self-administration
Whereas substitution procedures may be more sensitive for determining the reinforcing effects of a compound, acquisition procedures assess only the reinforcing effects of a single compound, without the potential confound of prior drug associations. The ability of MG and 7-HMG to engender and maintain responding without prior drug history was determined in a separate cohort of rats. Rats were assigned to one of three groups to self-administer MG (n = 8; 100 μg/infusion), 7-HMG (n = 8; 10 μg/infusion) or morphine (n = 6; 100 μg/infusion). Fol- lowing stable responding at the initial dose, rats in the MG group were given access to 25 and 50 μg/infusion MG, 7-HMG group was allowed to self-administer 5.0 and 2.5 μg/infusion 7-HMG, while rats in the morphine group were allowed to self-administer 50 μg/infusion morphine. The presentation of doses for MG and 7-HMG was randomized following the initial dose.
Experiment 3: Selective opiate receptor antagonism of morphine and 7-HMG self-administration
To determine the contribution of μ and δ opiate receptors on the reinforcing effects of 7-HMG, rats that had previ- ously acquired morphine and 7-MHG self-administration were administered naloxonazine (NLXZ), a selective μ1 receptor antagonist or the δ receptor antagonist naltrindole (NTI). NLXZ (5 and 15 mg/kg, i.p.) and NTI (0.5 and 5 mg/kg, i.p.) were administered 30 minutes before the session. The effects of saline pretreatment and saline extinction on responding were also assessed.
Data analysis
Experiment 1
Morphine self-administration as well as MG and 7-HMG substitution was analyzed using a one-factor ANOVA (Dose). Morphine self-administration before and after MG and 7-HMG substitution was analyzed using a two- way ANOVA (Dose × Pre/Post). Analysis of three days fol- lowing MG and 7-HMG substitution was conducted using a two-way repeated measures ANOVA (Dose × Pre/Post) with Sessions as the repeated measure.
Experiment 2
Acquisition of self-administration was analyzed using one-factor ANOVA (Dose) ANOVA and number of infu- sions as the dependent measure.
Experiment 3
Com- parisons between baseline intake and intake following saline pretreatment were conducted using two-tailed paired t-test for both the morphine and 7-HMG groups. The effects of NLXZ and NTI on morphine and 7-HMG were analyzed independently using a one-factor ANOVA (Antagonist Dose).
Analysis of the session in which the antagonists or saline were administered, and the follow- ing session was conducted using a two-way repeated measures ANOVA (Dose × Day) with Sessions as the repeated measure. The number of infusions was the dependent variable for analyses. Where appropriate, post hoc analyses were conducted using Tukey’s test.
RESULTS
Substitution and reintroduction of morphine
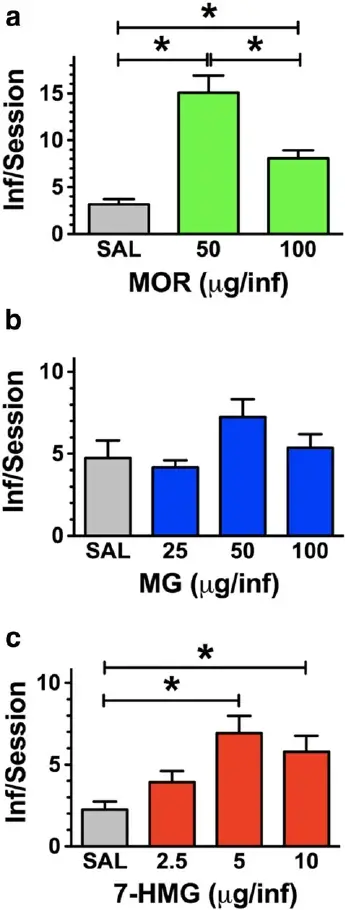
Responding was engendered and maintained by mor- phine at the doses tested [F(2,17) = 24.4, P < 0.0001]. Morphine self-administration was dose-dependent with both morphine doses significantly greater than saline and 50 μg greater than 100 μg/infusion (P < 0.05) (Fig. 1a). MG was not reliably self-administered when substituted for morphine [F(4,38) = 0.51, P = 0.73]. MG intake was not significantly different from vehicle, suggesting that MG does not function as a reinforcer at the doses tested (Fig. 1b). In contrast, 7-HMG substituted for morphine with intake dependent on the dose available [F(4,32) = 6.3, P = 0.0009]. The number of infusions obtained for 5 and 10 μg/inf were significantly greater than vehicle (P < 0.05), confirming that these doses of 7-HMG functioned as reinforcing stimuli. 7-HMG resulted in an inverted ‘U’-shaped dose-effect function with maxi- mal intake observed at 5 and 10 μg/inf (Fig. 1c).
Following completion of the substitution protocol, morphine self-administration was re-assessed. Morphine intake was significantly altered by both MG [F(1,32) = 5.5, P = 0.025] and 7-HMG [F(1,24) = 12.92, P = 0.0015], albeit in opposite direc- tions. MG exposure significantly reduced morphine self- administration of 50 μg (P < 0.01), but not 100 μg [Fig. 1d (top panel)]. The decrease in morphine self- administration (50 μg) was not observed on the first day following MG exposure but was significantly de- creased on days two and three Fig. 1c (middle panel)].